Major lead deposits are to be found in the recycling industry.
In the not too far distant future lead mineral deposits will have disappeared and the metal will only be found in scrap metal, mainly in used lead acid batteries.
This ‘lead deposit’ is vast and practically inexhaustible.
More than a billion vehicles are on the road of the planet and each vehicle is fitted with a starter battery.
The battery’s life is determined by its use:
![]() Number of start cycles
Number of start cycles
![]() Age of the vehicle
Age of the vehicle
![]() Local climate
Local climate
![]() Local road conditions
Local road conditions
![]() Vehicle maintenance
Vehicle maintenance
The life of a battery is variable from 2 to 6 years. If you take 4 years as an average you can easily see the importance of the ‘deposit’.
One billion vehicles = 250,000,000 batteries with an average weight of 20 kg per battery.
It is 5,000,000 tons of used batteries that we must recycle to respect the environment (3,100,000 tons of lead).
Battery recycling protects our planet, creates jobs and generates wealth.
Recycle batteries is protecting the planet, and creating jobs, but also $ 6.2 billion
Lead-acid batteries are infinitely recyclable.
If batteries represent an important lead deposit on the planet, it is also the most important source of its pollution.
Batteries are toxic but perfectly recyclable and safe with the modern equipment that has been developed by specialized industry and battery manufacturers who find in recycling the raw material they need.
2022 Dross Engineering presents a new furnace for the recovery of lead from dross
Electric rotary furnace with a capacity of 150 dm3 to treat dross and recover lead. The system is designed for dross producers such as battery, anode, strip manufacturers and secondary metal refiners.
This innovative furnace (patented) has a thermomechanical device that separates free lead from oxides. Highly efficient industrial system.
Recovered lead is acceptable for reuse
The lead recovery system allows battery producers to:
Dross Engineering, is one of the few manufacturers to design and build industrial equipment only for the recycling of non-ferrous scrap (lead-aluminium-zinc copper)
Over the years, (50 years in 2020) Dross Engineering has developed several innovative systems having been the subject of industrial protection.
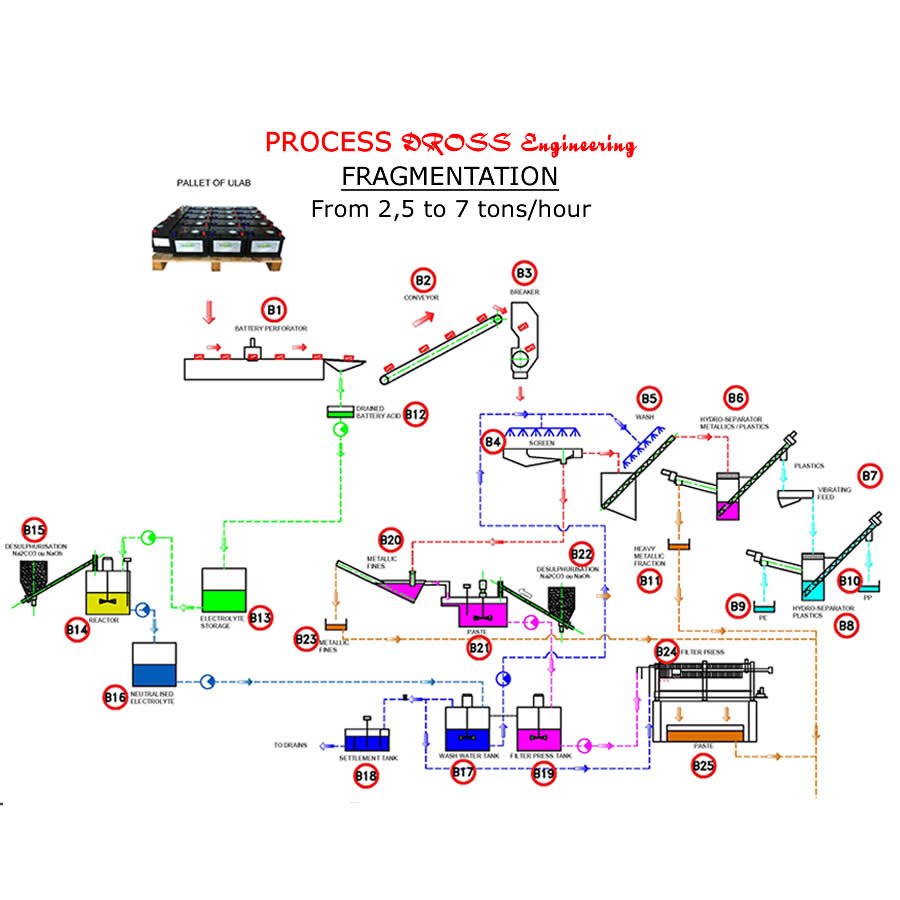
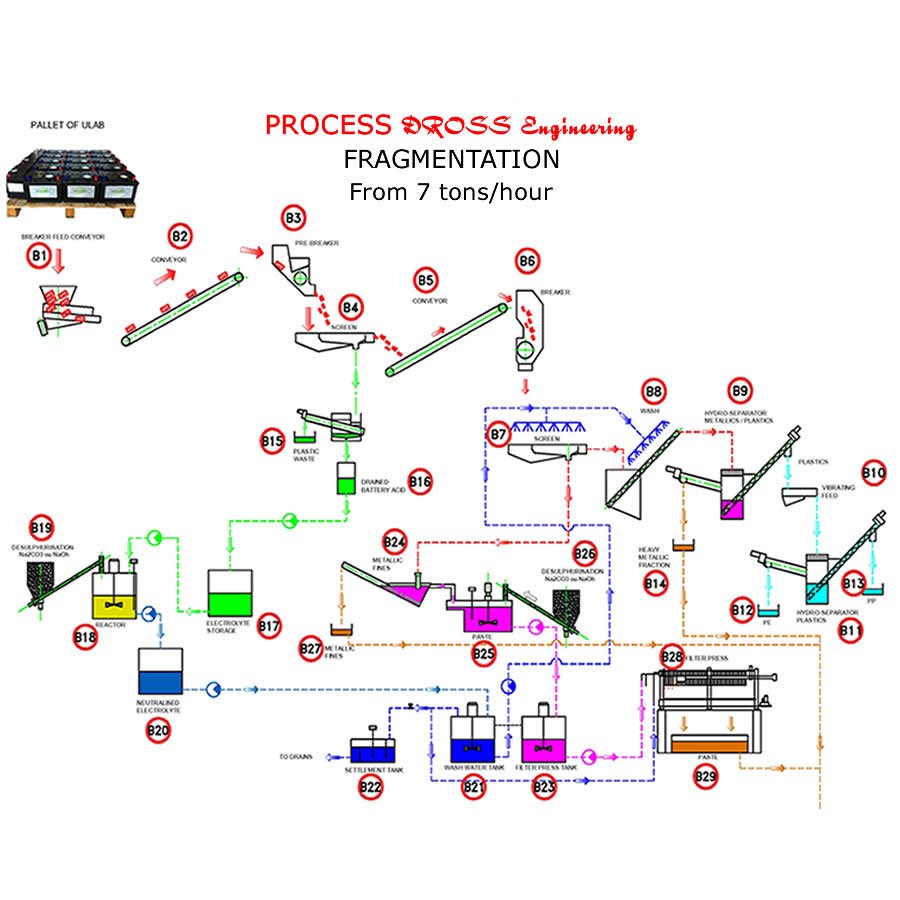
The Dross Engineering breaking lines allow the battery to be separated into 6 products instead of 4 in the past.
In old installations, the battery was crushed, treated with electrolysis and it is found at the exit of the paste (oxides and lead sulphate) containing 7 to 10% of sulfuric acid (So2 in the fumes), metal, polypropylene and acid-filled polyethylene
With the Dross Engineering process the electrolysis is recovered for recycling or neutralization.
Two categories of metal:
![]() Fine (particles smaller than 5 mm containing oxides) to be treated in a rotary furnace.
Fine (particles smaller than 5 mm containing oxides) to be treated in a rotary furnace.
![]() Large (terminal-grids and large pieces of lead without oxide) to be melted in kettle.
Large (terminal-grids and large pieces of lead without oxide) to be melted in kettle.
![]() A paste without metal (with very little copper-antimony-tin).
A paste without metal (with very little copper-antimony-tin).
![]() Polypropylene (casing).
Polypropylene (casing).
![]() Polyethylene (interlayer).
Polyethylene (interlayer).
The desulfurization process developed by Dross Engineering now allows:
Reduce reduction and refining times by saving a lot of energy and respecting the environment.
5 models 2.5t/h - 5t/h – 10t/h – 15t/h – 20t/h
Sulfur = 0.2% in the paste after desulfurization.
Ph = 6/7 for effluents.
Pb = 0.5mg / l of wastewater discharge.
17 breaking units using the Dross Engineering process are in operation and 2 in assembly on July 31, 2019.
The figures reported were recorded in Casablanca. The facility visited by the Ministry of the Environment of the Kingdom of Morocco.
The process works continuously and uses the choice of Na2CO3 or NaOh.
Consumption is largely compensated by savings during thermal operations to produce bullion metal.
Reduced consumption of Na2O3.
Less iron consumption.
Lower temperature in rotary furnaces = less gas consumption.
Smoke virtually without So2.
Lifetime of refractory increased.
A process designed to be offered at a price allowing its return on investment of less than two years.
Installation and commissioning by Dross Engineering.